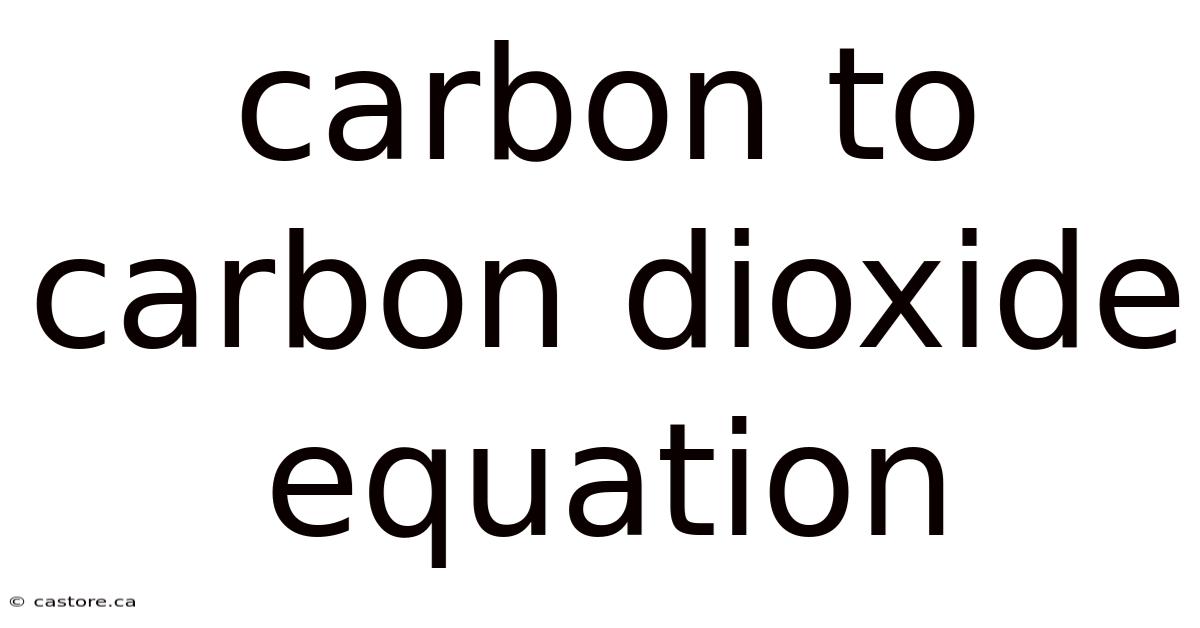Imagine a frosty winter morning. That simple act is a miniature illustration of the grand, continuous cycle of carbon, constantly transforming and circulating throughout our planet. In practice, as you exhale, you see a visible puff of breath—a fleeting cloud dissipating into the air. At its heart lies the seemingly simple, yet profoundly important, carbon to carbon dioxide equation, a fundamental process that underpins life as we know it and drives many of the Earth’s essential systems.
Consider the burning of a log in a fireplace. Think about it: the crackling flames and radiating heat are not just cozy comforts; they are a visible manifestation of a chemical transformation. This union births carbon dioxide, an invisible gas that quietly ascends into the atmosphere, carrying with it the energy that once bound the tree's very structure. The solid carbon within the wood, painstakingly built by the tree over decades through photosynthesis, is rapidly combining with oxygen from the air. Understanding the nuances of this carbon to carbon dioxide equation is not merely an academic exercise, but a crucial step in comprehending the complex dance of life, energy, and climate that defines our world.
Unveiling the Carbon to Carbon Dioxide Equation
The carbon to carbon dioxide equation represents a fundamental chemical reaction wherein carbon (C) atoms combine with oxygen (O₂) molecules to form carbon dioxide (CO₂). This process is often represented by the following balanced chemical equation:
C + O₂ → CO₂
At its core, this equation illustrates a process of oxidation. Here's the thing — oxygen gains these electrons, resulting in the formation of the stable molecule, carbon dioxide. While seemingly straightforward, this reaction is key in various natural and industrial processes, impacting everything from respiration and combustion to climate change. And carbon, in its elemental form or as part of a compound, loses electrons as it bonds with oxygen. The simplicity of the equation belies its profound implications for life and the environment.
To fully grasp the significance, one must dig into the underlying chemistry and the context in which this reaction occurs. Carbon, with its unique ability to form stable bonds with itself and other elements, is the backbone of organic molecules, the building blocks of all known life. Oxygen, a highly reactive element, is abundant in the atmosphere and essential for respiration in most living organisms. When these two elements meet under the right conditions, the result is the release of energy and the creation of carbon dioxide, a greenhouse gas that plays a critical role in regulating Earth's temperature.
Delving Deeper: Definitions, Scientific Foundations, and Essential Concepts
Carbon (C): A non-metallic element with atomic number 6. It exists in various allotropic forms, including diamond, graphite, and fullerenes. Carbon's ability to form four covalent bonds makes it uniquely suited to create complex molecules, the foundation of organic chemistry.
Oxygen (O₂): A diatomic molecule essential for respiration in most living organisms and a key component of combustion processes. It's a highly reactive element, readily combining with other elements to form oxides.
Carbon Dioxide (CO₂): A colorless, odorless gas composed of one carbon atom and two oxygen atoms. It is a greenhouse gas, meaning it traps heat in the atmosphere. CO₂ is a product of respiration, combustion, and decomposition, and is used by plants during photosynthesis.
Oxidation: A chemical reaction involving the loss of electrons by a molecule, atom, or ion. In the carbon to carbon dioxide equation, carbon is oxidized as it loses electrons to oxygen And that's really what it comes down to..
Combustion: A chemical process involving rapid reaction between a substance with an oxidant, usually oxygen, to produce heat and light. Burning wood, fossil fuels, and other organic materials are examples of combustion processes that release carbon dioxide.
Respiration: The process by which living organisms convert glucose and oxygen into energy, releasing carbon dioxide and water as byproducts. This is the reverse of photosynthesis Not complicated — just consistent..
Photosynthesis: The process by which plants and some other organisms use sunlight to synthesize foods from carbon dioxide and water. Photosynthesis removes carbon dioxide from the atmosphere.
Greenhouse Gas: A gas in the atmosphere that absorbs and emits radiant energy within the thermal infrared range. Greenhouse gases trap heat and contribute to the warming of the planet. Carbon dioxide is a major greenhouse gas Surprisingly effective..
The history of understanding the carbon to carbon dioxide equation is intertwined with the development of modern chemistry. So in the late 18th century, Antoine Lavoisier, considered the father of modern chemistry, conducted experiments that demonstrated the role of oxygen in combustion and respiration. He showed that burning carbon in oxygen produced carbon dioxide, laying the foundation for understanding the chemical equation as we know it today. Later, scientists like Svante Arrhenius recognized the potential of carbon dioxide to influence Earth's temperature, foreshadowing the modern understanding of climate change.
Real talk — this step gets skipped all the time Not complicated — just consistent..
Understanding the concepts surrounding the carbon to carbon dioxide equation requires appreciating its role in the carbon cycle, a biogeochemical cycle that describes the continuous movement of carbon between the atmosphere, oceans, land, and living organisms. Carbon dioxide is a key component of this cycle, entering the atmosphere through respiration, combustion, and volcanic eruptions, and being removed through photosynthesis and absorption by the oceans. Human activities, particularly the burning of fossil fuels, have significantly altered this cycle, leading to an increase in atmospheric carbon dioxide concentrations and contributing to global warming.
To build on this, the energy dynamics of the carbon to carbon dioxide equation are crucial. Think about it: the reaction is exothermic, meaning it releases energy in the form of heat and light. This energy release is what powers combustion engines and provides warmth when burning fuels. On the flip side, the amount of energy released depends on the specific carbon-containing compound being oxidized and the efficiency of the combustion process. Incomplete combustion can lead to the formation of other carbon-containing compounds, such as carbon monoxide (CO), a toxic gas.
Finally, it’s important to note that the carbon to carbon dioxide equation is not just a theoretical concept. It’s a process that occurs constantly and in vast quantities throughout the world. From the cellular respiration in our bodies to the burning of forests in wildfires, the conversion of carbon to carbon dioxide is a fundamental aspect of life and the Earth’s environment Less friction, more output..
Trends and Latest Developments
Current trends surrounding the carbon to carbon dioxide equation are heavily influenced by concerns about climate change and the increasing concentration of CO₂ in the atmosphere. Scientists and policymakers are focused on understanding the sources and sinks of carbon dioxide, as well as developing strategies to reduce emissions and mitigate the impacts of climate change Nothing fancy..
One major trend is the increased monitoring and measurement of carbon dioxide levels globally. Scientists use various techniques, including satellite observations, ground-based sensors, and oceanographic surveys, to track the movement and concentration of CO₂ in different parts of the Earth system. This data is crucial for understanding the effectiveness of climate mitigation efforts and for predicting future climate scenarios.
It sounds simple, but the gap is usually here Worth keeping that in mind..
Another important trend is the development of carbon capture and storage (CCS) technologies. CCS aims to capture carbon dioxide emissions from industrial sources, such as power plants and cement factories, and store them underground or in other long-term storage solutions. While CCS is still in its early stages of development, it has the potential to significantly reduce carbon dioxide emissions from key sectors of the economy.
Beyond that, there is growing interest in carbon utilization technologies, which aim to convert captured carbon dioxide into valuable products, such as fuels, chemicals, and building materials. This approach not only reduces carbon dioxide emissions but also creates economic opportunities.
The scientific community is also actively researching natural carbon sinks, such as forests and oceans, to understand their capacity to absorb carbon dioxide from the atmosphere. Protecting and enhancing these natural sinks is a crucial part of climate mitigation efforts. As an example, reforestation projects can help to sequester carbon dioxide from the atmosphere and store it in biomass.
Some disagree here. Fair enough.
Public opinion and policy are also evolving. That's why there is increasing public awareness of the risks of climate change and growing support for policies that reduce carbon dioxide emissions. Governments around the world are implementing various policies, such as carbon taxes, emissions trading schemes, and renewable energy mandates, to incentivize the transition to a low-carbon economy. The Paris Agreement, an international accord aimed at limiting global warming, reflects a global commitment to reducing carbon dioxide emissions and addressing climate change.
Professional insights suggest that addressing the challenges related to the carbon to carbon dioxide equation requires a multi-faceted approach. This includes reducing emissions from fossil fuels, developing and deploying carbon capture and utilization technologies, protecting and enhancing natural carbon sinks, and implementing effective climate policies. Collaboration between scientists, policymakers, businesses, and the public is essential to achieving meaningful progress.
Tips and Expert Advice
Understanding the carbon to carbon dioxide equation is not just for scientists and policymakers; it can also inform our everyday choices and actions. Here are some practical tips and expert advice for reducing your carbon footprint and contributing to a more sustainable future:
1. Reduce Your Energy Consumption: Energy production, especially from fossil fuels, is a major source of carbon dioxide emissions. By reducing your energy consumption, you can directly lower your contribution to these emissions.
- Use energy-efficient appliances: When purchasing new appliances, look for Energy Star certified models. These appliances use significantly less energy than standard models.
- Switch to LED lighting: LED bulbs use up to 75% less energy and last much longer than incandescent bulbs.
- Unplug electronics when not in use: Many electronic devices continue to draw power even when turned off. Unplugging them can save energy and reduce your electricity bill.
- Adjust your thermostat: Lowering your thermostat in the winter and raising it in the summer can significantly reduce your energy consumption for heating and cooling.
- Improve insulation: Proper insulation can help to keep your home warm in the winter and cool in the summer, reducing the need for heating and cooling.
2. Choose Sustainable Transportation Options: Transportation is another major source of carbon dioxide emissions. By choosing more sustainable transportation options, you can significantly reduce your carbon footprint Not complicated — just consistent. Which is the point..
- Walk or bike: Walking or biking is not only good for your health but also produces zero carbon dioxide emissions.
- Use public transportation: Buses and trains are more energy-efficient than individual cars, especially when they are full.
- Carpool: Sharing a ride with others can reduce the number of cars on the road and lower carbon dioxide emissions.
- Drive an electric or hybrid vehicle: Electric vehicles produce zero tailpipe emissions, and hybrid vehicles use less fuel than traditional gasoline-powered cars.
- Drive efficiently: Avoid aggressive driving, maintain proper tire pressure, and keep your car well-maintained to improve fuel efficiency.
3. Reduce, Reuse, and Recycle: The production and disposal of goods consume energy and resources, leading to carbon dioxide emissions. By reducing, reusing, and recycling, you can minimize these emissions Turns out it matters..
- Reduce consumption: Buy less stuff. Before making a purchase, ask yourself if you really need it.
- Reuse items: Find new uses for old items instead of throwing them away.
- Recycle: Recycle paper, plastic, glass, and metal. Recycling reduces the need to extract and process raw materials, which saves energy and reduces carbon dioxide emissions.
- Compost: Composting food scraps and yard waste reduces the amount of organic waste sent to landfills, where it decomposes and releases methane, a potent greenhouse gas.
4. Eat Sustainably: The food we eat also contributes to carbon dioxide emissions, particularly through agriculture and transportation. By making sustainable food choices, you can reduce your carbon footprint.
- Eat less meat: Meat production, especially beef, is a major source of greenhouse gas emissions.
- Buy local and seasonal food: Buying food that is grown locally and in season reduces the need for transportation and refrigeration, which saves energy.
- Reduce food waste: About one-third of the food produced globally is wasted. Reducing food waste can save energy and resources.
- Grow your own food: Growing your own fruits, vegetables, and herbs can reduce your reliance on the industrial food system.
5. Advocate for Change: Individual actions are important, but systemic change is also needed to address climate change. By advocating for policies that reduce carbon dioxide emissions, you can have a greater impact Turns out it matters..
- Support renewable energy: Advocate for policies that promote the development and deployment of renewable energy sources, such as solar, wind, and geothermal.
- Support carbon pricing: Advocate for carbon taxes or emissions trading schemes, which incentivize businesses and individuals to reduce carbon dioxide emissions.
- Support energy efficiency standards: Advocate for policies that require buildings and appliances to meet certain energy efficiency standards.
- Vote for climate-friendly candidates: Support political candidates who prioritize climate action.
FAQ
Q: What is the chemical formula for carbon dioxide?
A: The chemical formula for carbon dioxide is CO₂. This indicates that each molecule of carbon dioxide consists of one carbon atom and two oxygen atoms Small thing, real impact..
Q: Is the reaction of carbon to carbon dioxide endothermic or exothermic?
A: The reaction is exothermic, meaning it releases energy in the form of heat and light. This is why burning carbon-containing fuels produces heat That alone is useful..
Q: What are the main sources of carbon dioxide emissions?
A: The main sources of carbon dioxide emissions include the burning of fossil fuels (coal, oil, and natural gas) for energy production, industrial processes (such as cement manufacturing), deforestation, and agriculture Took long enough..
Q: How does carbon dioxide contribute to climate change?
A: Carbon dioxide is a greenhouse gas, meaning it traps heat in the atmosphere. Increased concentrations of carbon dioxide in the atmosphere lead to a warming of the planet, resulting in climate change Worth keeping that in mind..
Q: What are some ways to reduce carbon dioxide emissions?
A: Ways to reduce carbon dioxide emissions include using energy-efficient appliances, switching to renewable energy sources, reducing meat consumption, driving less, and recycling.
Q: What is carbon capture and storage (CCS)?
A: Carbon capture and storage (CCS) is a technology that captures carbon dioxide emissions from industrial sources and stores them underground or in other long-term storage solutions Easy to understand, harder to ignore..
Q: What is carbon utilization?
A: Carbon utilization is the process of converting captured carbon dioxide into valuable products, such as fuels, chemicals, and building materials.
Conclusion
The carbon to carbon dioxide equation, while seemingly simple, represents a fundamental chemical process with far-reaching implications for life on Earth. Which means from powering our bodies through respiration to driving climate change through greenhouse gas emissions, this equation is key here in shaping our world. Understanding the science behind it, as well as the trends and developments surrounding it, empowers us to make informed decisions and take meaningful action to reduce our carbon footprint.
By adopting sustainable practices in our daily lives, supporting policies that promote clean energy, and advocating for systemic change, we can collectively work towards a more sustainable future. We encourage you to explore the links provided, delve deeper into the science, and actively engage in efforts to mitigate climate change. This leads to share this article with your friends and family to spread awareness and inspire action. Together, we can make a difference in addressing the challenges posed by the carbon to carbon dioxide equation and building a healthier planet for future generations.